It has been found that an atom has a complex structure and is composed of sub-atomic particles such as electrons, protons and neutrons.Ītoms of the same element may not be similar in all respects. The main modifications made in this theory are as follows :Ītom is no longer considered to be indivisible. It did not make any distinction between ultimate particle of an element that takes part in reactions (atom) and ultimate particle that has independent existence (molecule). It failed to explain Gay Lussac’s law of combining volumes. It failed to explain the nature of forces that bind together different atoms in a molecule. It could not explain how and why atoms of different elements combine with each other to form compound-atoms or molecules.
It failed to explain how atoms of different elements differ from each other, i.e., it did not tell anything about structure of the atom.

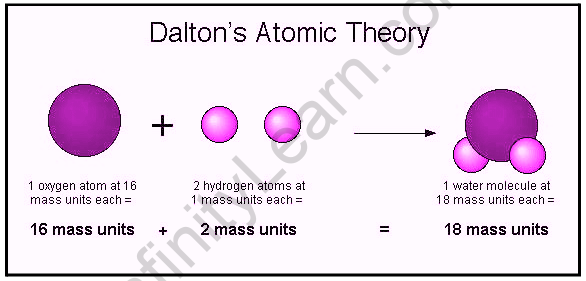
The main failures of Dalton’s atomic theory are : Matter is made up of extremely small, indivisible particles called atoms.Ītoms of same substance are identical in all respects, i.e., they possess same size, shape, mass, chemical properties, etc.Ītoms of different substances are different in all respects, i.e., they possess different sizes, shapes, masses, chemical properties, etc.Ītom is the smallest particle that takes part in chemical reactions.Ītoms of different elements may combine with each other in a fixed, simple, whole number ratio to form compound atoms.Ītoms can neither be created nor destroyed, i.e., atoms are indestructible. The main points of Dalton’s atomic theory are as follows:
